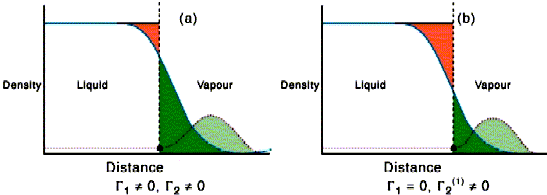
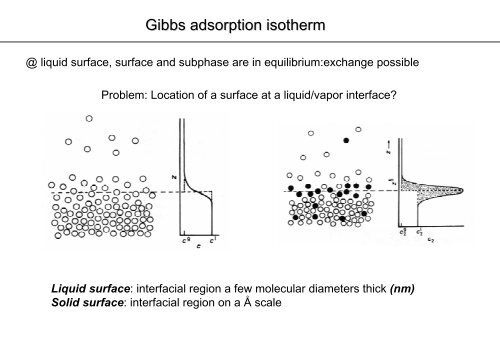
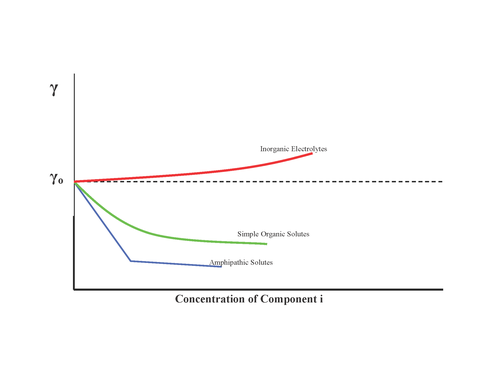
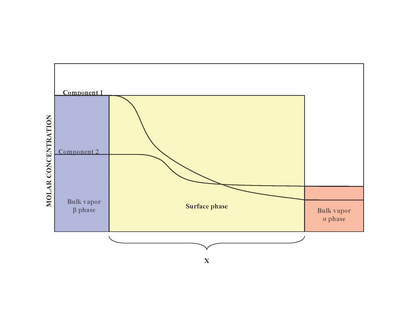
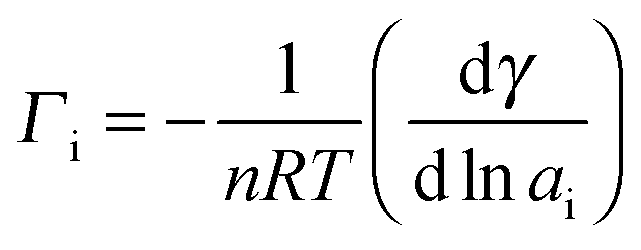Adsorption of geses on liquids. Surface-active and surface-inactive substances. Gibbs's equation, Shyshkovsky's equations and Langmuir's equations Plan. - ppt download

A) Gibbs adsorption isotherm of oLMD1/3/3 (c = 0.025 mg mL⁻¹) in 20 ×... | Download Scientific Diagram

Applicability of the Gibbs Adsorption Isotherm to the analysis of experimental surface-tension data for ionic and nonionic surfactants - ScienceDirect

Applicability of the Gibbs Adsorption Isotherm to the analysis of experimental surface-tension data for ionic and nonionic surfactants - ScienceDirect

d) Activated coconut charcudi 68. The equation. - is (a) Gibbs adsorption isotherm (b) Freundlich adsorption isotherm (c) Langmuir adsorption isotherm (d) BET equation The adsorntion isotherm a gas is given E 69.

8.4 Surface adsorption of solution. 1 The surface phenomena of solution: Is solution homogeneous? (1) surface adsorption AA A B A B Solvent A Solute. - ppt download

PDF) Extracting the equation of state of lattice gases from Random Sequential Adsorption simulations by means of the Gibbs adsorption isotherm
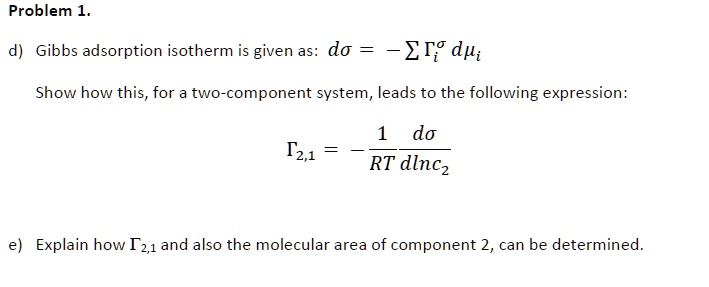
SOLVED: Gibbs adsorption isotherm is given as: dΓ = -SdT + VdP. Show how this, for a two-component system, leads to the following expression: dΓ = RT d(ln c1/c2) Explain how the

An examination of the one-parameter adsorption equation without using the Gibbs adsorption equation - ScienceDirect