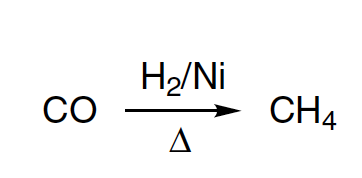lu) 2116 TPC15 25. An alkane with even number of carbon only, can result in: (a) Sabatier Senderens reaction (b) Wurtz reaction (C) Kolbe's electrolytic reaction (d) Grignard reaction 26 n.Heyane can
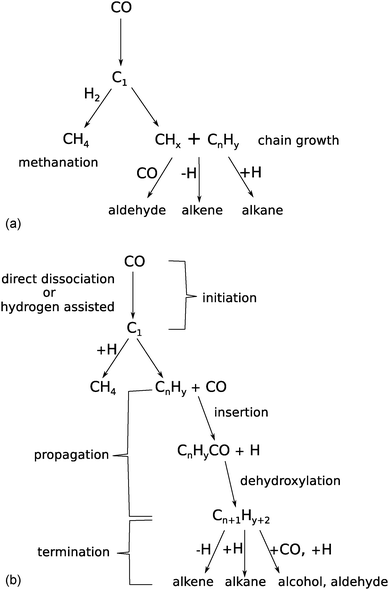
Mechanism and microkinetics of the Fischer–Tropsch reaction - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C3CP52506F
LIST - 1 LIST - II A) Sabatier Senderens reaction 1) Anhydrous AlCl 3 B) Friedel Crafts reaction 2) Ni/H 2 C) Wurtz reaction 3) Sodium acetate D) Kolbe's electrolysis 4) Na/Dry
LIST - 1 LIST - II A) Sabatier Senderens reaction 1) Anhydrous AlCl 3 B) Friedel Crafts reaction 2) Ni/H 2 C) Wurtz reaction 3) Sodium acetate D) Kolbe's electrolysis 4) Na/Dry
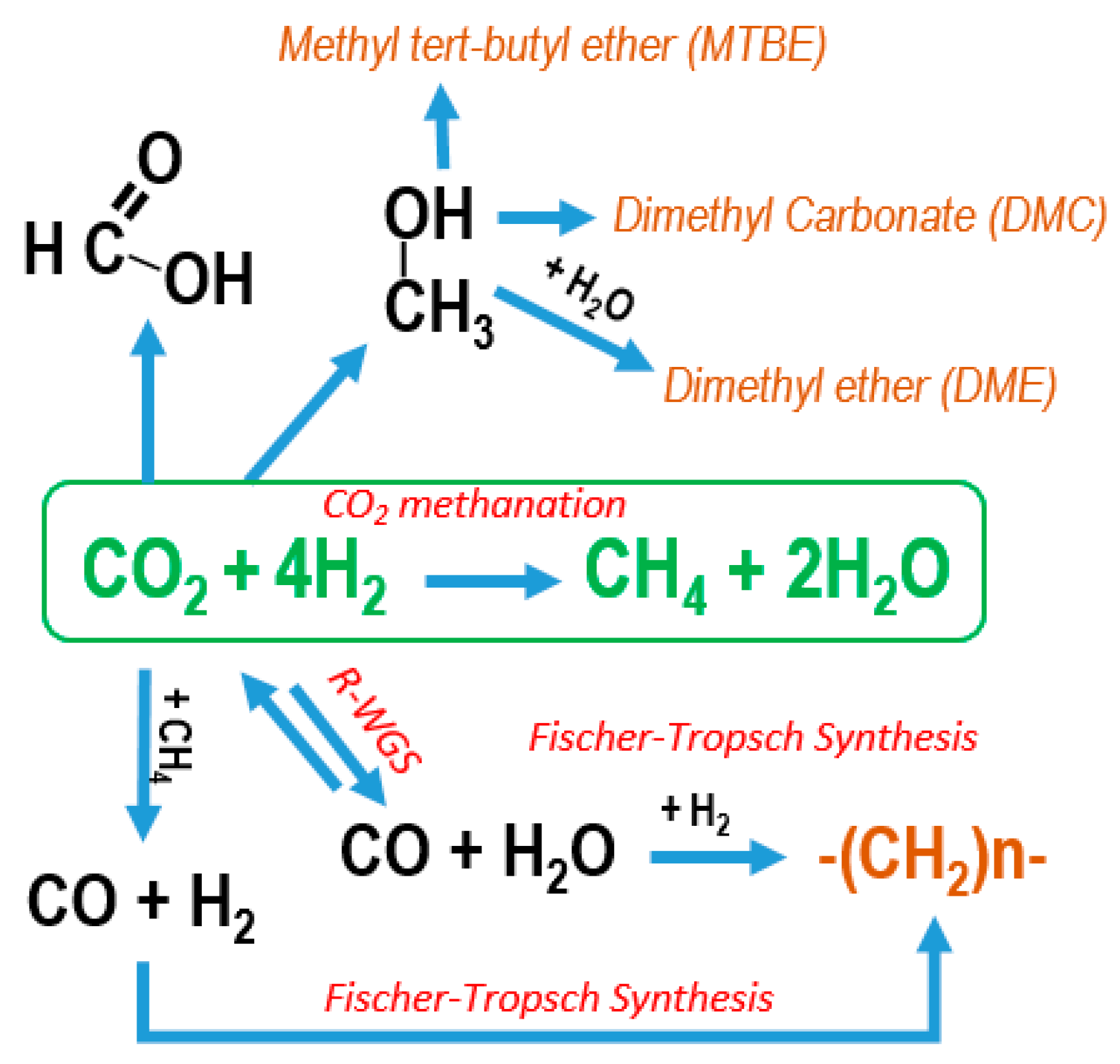
Catalysts | Free Full-Text | Policies and Motivations for the CO2 Valorization through the Sabatier Reaction Using Structured Catalysts. A Review of the Most Recent Advances
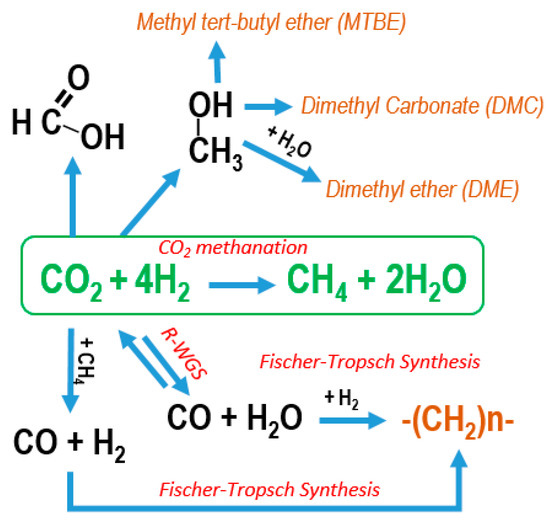
Catalysts | Free Full-Text | Policies and Motivations for the CO2 Valorization through the Sabatier Reaction Using Structured Catalysts. A Review of the Most Recent Advances

SOLVED: The reaction CH2=CH2+H2Ni / 250-300^∘C⟶CH3-CH3 is called: (a) Wurtz reaction (b) Kolbe's reaction (c) Carbylamine reaction (d) Sabatier-Senderens reaction

Ethane is obtained from ethylene by which of the following methods?a) Wurtz reaction b) Dehydrogenation c) Frakland reaction d) Sabatier-Senderens reduction Correct answer is option 'D'. Can you explain this answer? -
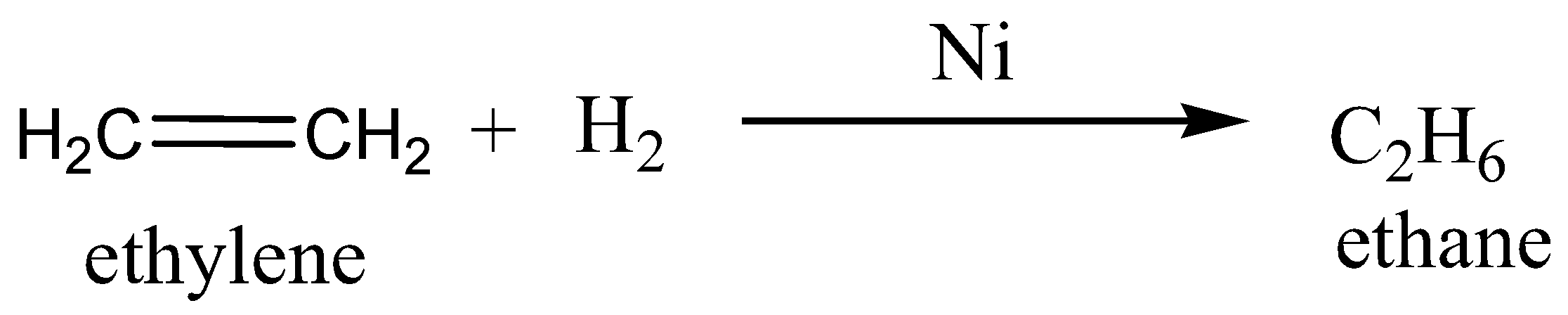
In which of the following reactions a new C-C bond is formed in the preparation of ethane?A) Sabatier-Senderens reaction.B) Reduction of ethyl iodide.C) Decarboxylation.D) Kolbe's electrolysis.